Akari Therapeutics Announces Positive Initial Phase II Clinical Data in Orphan Skin Disease Bullous Pemphigoid
NEW YORK and LONDON,
|
|||||
Bullous pemphigoid is a severe orphan inflammatory skin disease currently treated primarily with steroids and immunosuppressants which bring with them well known side effects. Treatment response and steroid potency varies significantly based on the severity of the disease, although flares and relapses frequently occur.
In patients with bullous pemphigoid there is evidence that both terminal complement activation (C5) and the lipid mediator leukotriene B4 (LTB4) have a central role in driving the disease. Ex vivo data, from a recent study at
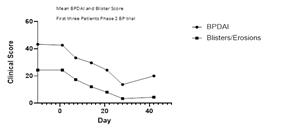
The Phase II trial for up to nine mild-to-moderate bullous pemphigoid patients is a six-week open-label single-arm study evaluating safety and with the main efficacy measure the Bullous Pemphigoid Disease Area Index (BPDAI) a frequently used evaluation of the extent and severity of the disease.
Initial results from the first three patients showed that Nomacopan (Coversin), dosed daily subcutaneously, was well tolerated in three elderly patients (>65 years), and that there were no drug-related adverse events.
Prior to treatment with Nomacopan (Coversin), two out of the three patients were already on topical corticosteroids (mometasone) while a third was naïve to steroid treatment. Steroids were reduced at weekly intervals so that by day 21 both patients were only treated with Nomacopan (Coversin). In the 7-11-day period prior to initiation on Nomacopan (Coversin), the two patients on steroids showed either no or minor improvement in their BPDAI global score (between 0% and 5%) and no improvement in blisters.
By Day 7, 21 and 42 of treatment with Nomacopan (Coversin), the BPDAI global score fell by a mean of 31%, 45% and 52%, respectively.
By Day 7, 21 and day 42 of treatment with Nomacopan (Coversin), blisters/erosions dropped by a mean of 45%, 75% and 87%, respectively.
“The initial Phase II data in BP patients treated with Nomacopan (Coversin) is very encouraging, indicating that BP can potentially be resolved without the adverse issues caused by current steroidal treatments,” said Professor Detlef Zillikens and Professor Christian Sadick – lead investigators –
A graph accompanying this announcement is available at http://www.globenewswire.com/NewsRoom/AttachmentNg/9b0b8fe9-681d-48b5-a02f-801559113cf4
Clive Richardson, interim CEO of Akari Therapeutics, said, “As a result of this encouraging data, we plan to expand the trial to include additional severe patients by way of an amendment. We believe this promising data helps validate our strategy of focusing on those poorly treated orphan diseases where both C5 and LTB4 are implicated.”
About Akari Therapeutics
Akari is a biopharmaceutical company focused on developing inhibitors of acute and chronic inflammation, specifically for the treatment of rare and orphan diseases, in particular those where the complement (C5) or leukotriene (LTB4) systems, or both complement and leukotrienes together, play a primary role in disease progression. Akari's lead drug candidate, Nomacopan (Coversin), is a C5 complement inhibitor that also independently and specifically inhibits leukotriene B4 (LTB4) activity. Nomacopan (Coversin) is currently being clinically evaluated in four indications: bullous pemphigoid (BP), atopic keratoconjunctivitis (AKC), thrombotic microangiopathy (TMA), and paroxysmal nocturnal hemoglobinuria (PNH). Akari believes that the dual action of Nomacopan (Coversin) on both C5 and LTB4 may be beneficial in AKC and BP. Akari is also developing other tick derived proteins, including longer acting versions.
Cautionary Note Regarding Forward-Looking Statements
Certain statements in this press release constitute “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. These forward-looking statements reflect our current views about our plans, intentions, expectations, strategies and prospects, which are based on the information currently available to us and on assumptions we have made. Although we believe that our plans, intentions, expectations, strategies and prospects as reflected in or suggested by those forward-looking statements are reasonable, we can give no assurance that the plans, intentions, expectations or strategies will be attained or achieved. Furthermore, actual results may differ materially from those described in the forward-looking statements and will be affected by a variety of risks and factors that are beyond our control. Such risks and uncertainties for our company include, but are not limited to: needs for additional capital to fund our operations, our ability to continue as a going concern; uncertainties of cash flows and inability to meet working capital needs; an inability or delay in obtaining required regulatory approvals for Nomacopan (Coversin) and any other product candidates, which may result in unexpected cost expenditures; our ability to obtain orphan drug designation in additional indications; risks inherent in drug development in general; uncertainties in obtaining successful clinical results for Nomacopan (Coversin) and any other product candidates and unexpected costs that may result therefrom; difficulties enrolling patients in our clinical trials; failure to realize any value of Nomacopan (Coversin) and any other product candidates developed and being developed in light of inherent risks and difficulties involved in successfully bringing product candidates to market; inability to develop new product candidates and support existing product candidates; the approval by the FDA and EMA and any other similar foreign regulatory authorities of other competing or superior products brought to market; risks resulting from unforeseen side effects; risk that the market for Nomacopan (Coversin) may not be as large as expected; risks associated with the departure of our former Chief Executive Officers and other executive officers; risks associated with the SEC investigation; inability to obtain, maintain and enforce patents and other intellectual property rights or the unexpected costs associated with such enforcement or litigation; inability to obtain and maintain commercial manufacturing arrangements with third party manufacturers or establish commercial scale manufacturing capabilities; the inability to timely source adequate supply of our active pharmaceutical ingredients from third party manufacturers on whom the company depends; unexpected cost increases and pricing pressures and risks and other risk factors detailed in our public filings with the
For more information
Investor Contact:
Peter Vozzo
(443) 213-0505
peter.vozzo@westwicke.com
Media Contact:
Sukaina Virji / Nicholas Brown / Lizzy Seeley
+44 (0)20 3709 5700
Akari@consilium-comms.com
Source: Akari Therapeutics Plc